Despite our best efforts, chemicals with anthropogenic origin continue to be released into the environment, often with unquantified deleterious consequences. On the one hand, the lack of cost-effective strategies for water treatment and site remediation of conventional contaminants remains a major impediment to the protection and restoration of environment systems. On the other hand, the emerge of various new categories of contaminants such as per- and polyfluoroalkyl substances (PFASs) in recent years as a result of the advances in instrumentation poses serious challenges in the development of remediation and destruction technologies. Thus, we effort in the analysis, environmental fate, and degradation of organic contaminants, particularly the emerging ones. In combination with DFT, we illustrate their reaction and transformation mechanisms during degradation tests.
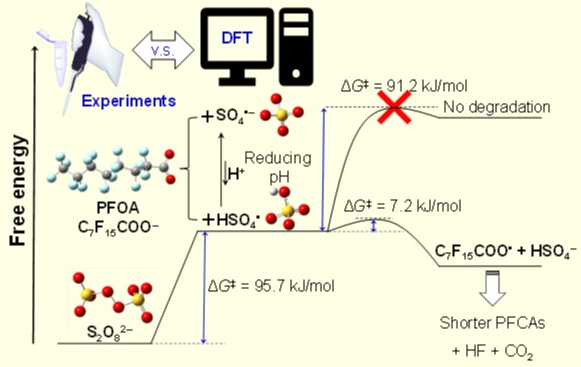
Peroxydisulfate (PDS) is one of the most commonly used agents for in situ chemical oxidation. Sulfate radical generated from thermally activated PDS can completely degrade perfluorooctanoic acid (PFOA) into carbon dioxide and fluoride ions but only at pH below 3. We mapped the entire reaction path of PFOA degradation by sulfate radical and explained the demand for low pH using density functional theory (DFT) calculations. The validity of the DFT methods was verified by the consistency of the free energy of activation derived theoretically with experimental data. The results provide valuable insight into remediation strategies including PDS as an oxidizing agent for PFASs. (Zhang et al., ES&T, 2019)
Co(ll) activated peroxymonosulfate (Co(ll)/PMS) generates sulfate radical at room temperature. However, we found that Co(ll)/PMS was barely reactive to PFOA but simultaneously transformed 6:2 fluorotelomer sulfonate (6:2 FTSA) to PFCAs of different chain length (C2-C7). Hydroxyl radical, instead of the expected sulfate radical, was confirmed to be the oxidant. This is the first study that demonstrates hydroxyl radical in Co(ll)/PMS systems can play a significant role in contaminant transformations. DFT calculations resolved the entire transformation pathways and found that the critical step was the formation of ketone radicals after oxygen addition. The results show how hydroxyl radical would adversely affect PFASs that contain aliphatic carbons in natural and engineered systems. (Zhang et al., ES&T, 2020; Zhang et al., ES&T, 2021)

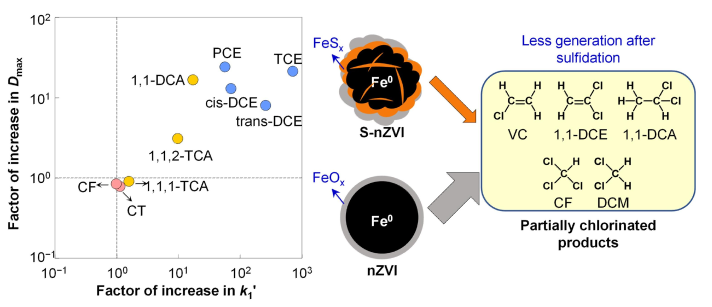
Chlorinated hydrocarbons (CHCs) have been widely used in various industry and are prevalent in most contaminated groundwater, which threatens the water supply of half the world population. Most CHCs form nonaqueous phase liquids at the bottom of the aquifers, and thus require in situ source zone treatments. Sulfidated nanoscale zerovalent iron (S-nZVl) is considered as the most promising agents because of the high reactivity, low cost, and less adverse impact on the ecosystem. We found that sulfidation not only enhances the reactivity of most CHCs, but also promotes the degradation pathways leading to the non-chlorinated benign products. The results demonstrate the advantages and feasibility of S-nZVl for in situ remediation of priority chlorinated solventsand contribute significantly to the knowledge on their dichlorination mechanisms. (Zhang et al., WR, 2021; Garcia#, Zhang#, et al., ES&T, 2021)
PFASs are often encountered together with CHCs in groundwater at sites affectedby firefighting activities. Our data show that nanoscale zerovalent iron (nZVl), sulfidated S-nZVl, and Pd-nZVI cannot degrade perfluoroalkyl acids (PFAAs), the most persistent PFASs, and thus break the illusion of using functionalized iron to treat PFAAs. However, PFAAs showed strong sorptive affinity for nZVl, which was not affected by the changes in surface chemistry with aging. The results suggest that injection of nZVl would not have negative effect on PFAA contamination in groundwater, instead, it could reduce PFAA concentrations near the remediation points. (Zhang et al., ES&T, 2018)

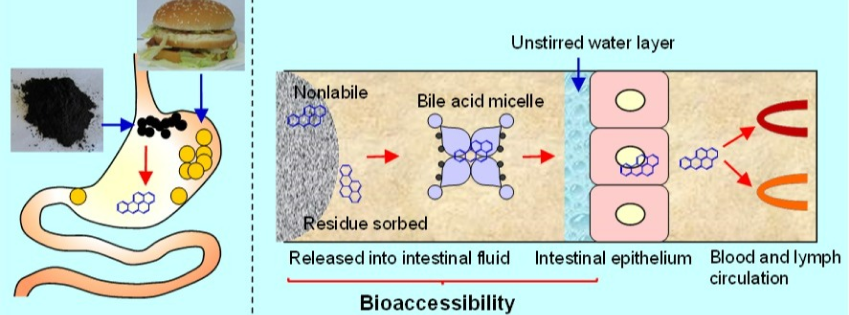
Polycyclic aromatic hydrocarbons (PAHs) are generated from incomplete combustion of carbonaceous materials such as fossil fuels and biomass and area well-known class of carcinogens. Surface soil is the major sink for PAHs, and PAHs in soil are strongly associated with soot materials. The unintentional ingestion of soil or soot particles is an important exposure pathway of PAHs especially for small children. For the first time, we assessed the gastrointestinal bioaccessbility of PAHs in soot and the impact from changing in physiological conditions associated with food ingestion and aging of soot. The data provide solid evidences on why risk assessment of organic contaminants need toconsider the nonlabile fraction in environmental particles and the variability in bioaccessibility caused by co-consumption of food. (Zhang et al., ES&T, 2015a; 2015b)